VUMC to help engage more study participants in research decisions
Human research will be more equitable, inclusive and stronger if the people who are participants in studies also help plan and guide the direction of those studies.
Human research will be more equitable, inclusive and stronger if the people who are participants in studies also help plan and guide the direction of those studies.

muhamed.baljevic@vumc.org
jennifer.choe@vumc.org

ryan.belcher@vumc.org
2026 V-EXCEL program dates: May 26, 2026 - July 31, 2026
For those interested in applying for the 2026-2028 VERTICAL Program, please complete an interest form here by December 1, 2025 at 11:59 PM CST. Leveraging the strengths of the Vanderbilt-Ingram Cancer Center, Vanderbilt University, and Vanderbilt University Medical Center, this program will immerse eight interns in a research laboratory for 10 weeks working with a Vanderbilt faculty member on a cutting-edge cancer research project.
Selected participants will receive a competitive stipend to support housing and living expenses. Through this internship, participants will gain the knowledge, skills, and community to further their education and career in cancer research or medicine. In addition to their research, interns will participate in:
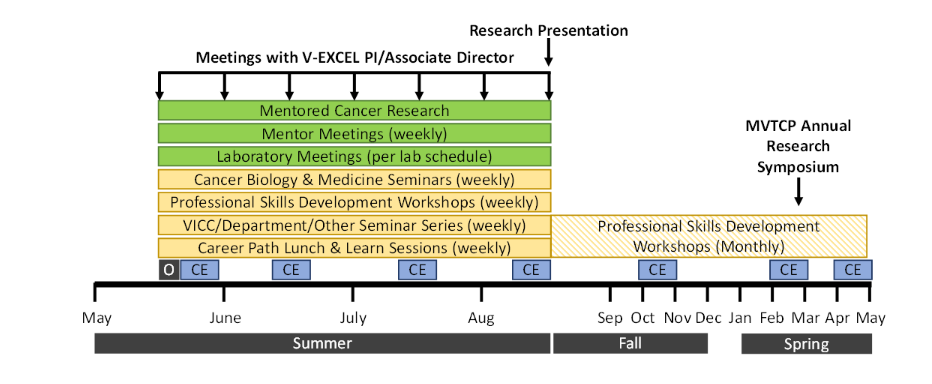
The activities after the summer session are not required. These include the Professional Skills Development Workshops, the MVTCP Annual Research Symposium, and Community Engagement (CE) events.
Eligible candidates for the V-EXCEL program will be:
The 2026 summer application deadline is December 1, 2025 at 11:59 pm CST. The following Materials are required:
Apply here: 2026 V-EXCEL Application
Please contact us at V-EXCEL@vumc.org.

janet.markle@vumc.org