Study led by Monroe Carell Jr. Children’s Hospital at Vanderbilt looks at international cohort with rare cancer
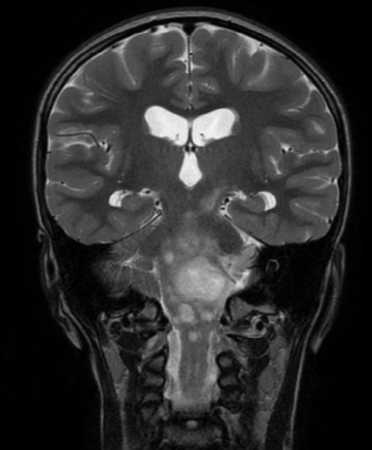
Pediatric-type, low-grade gliomas (PLGG) are the most common central nervous system (CNS) tumor in children, but in rare cases the tumors spread — known as disseminated pediatric low-grade gliomas (DPLGGs) — which leads to increased morbidity and mortality.
Researchers, led by Michael Dewan, MD, MSCI, compiled an international cohort of more than 260 DPLGG patients from 39 sites and 13 countries to study why low-grade brain tumors behave aggressively, as well as to identify the causative genetic alteration. Study findings were recently published in Neuro-Oncology.
“Our findings will help predict which children are at an elevated risk of developing disseminated low-grade gliomas as well as serve as a guide toward a more effective, personalized treatment,” said Dewan, associate professor of Neurological Surgery and Pediatrics at Monroe Carell Jr. Children’s Hospital at Vanderbilt and one of the study’s senior authors.
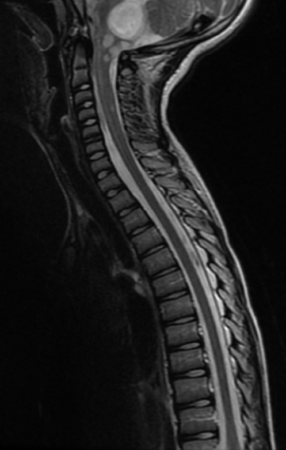
“Not all childhood brain tumors that look low-grade act the same,” he said. “This study expands our understanding of the clinical, pathologic and molecular features of this challenging disease. We found that when these tumors spread early or widely — especially in young children — outcomes are worse, but newer, targeted drugs work better than standard chemotherapy for many patients.”
Study findings inform prognosis and treatment decisions, particularly by supporting the earlier use of targeted, less-invasive therapies that may enhance survival and reduce treatment toxicity for children.
“This research improves our clinical and biological understanding of this rare disease, provides insights for improving patient care, and directs future clinical trials and basic science research,” said Dewan.
The post Study led by Monroe Carell Jr. Children’s Hospital at Vanderbilt looks at international cohort with rare cancer appeared first on Vanderbilt Health News.
Vanderbilt Health and Bertis establish collaboration for cancer drug discovery
Vanderbilt Health and Bertis, an artificial intelligence-driven proteomics-based precision medicine company, have announced a joint research and co-development collaboration. The endeavor marks a significant milestone in oncology by advancing the convergence of AI, spatial biology and translational cancer research.
By integrating Vanderbilt Health’s Molecular AI Initiative capabilities with Bertis’ proprietary deep proteomics and AI-enabled target discovery technologies, the collaboration will build an advanced, spatially resolved dataset to identify novel therapeutic targets and predictive biomarkers.
Traditional target discovery often relies on bulk tissue analysis, which loses the critical context of how cells are organized within a tumor. Vanderbilt Health’s Molecular AI approach changes this paradigm by employing sophisticated computational spatial analysis to generate high-resolution spatial molecular maps. This AI-driven spatial biology allows researchers to visualize and decode the complex architecture of the tumor microenvironment, specifically identifying how tumor, immune and stromal (connective tissue) cells interact in biologically and therapeutically relevant regions. By mapping the precise locations and spatial relationships of these cells, the Molecular AI platform can isolate the key cell populations responsible for treatment response or resistance.
These advanced spatial insights are then integrated with Bertis’ cutting-edge proteomics capabilities. While Vanderbilt Health maps the critical spatial context, Bertis will conduct deep proteomic and metabolomic profiling, applying its proprietary AI-enabled computational models to prioritize the most viable, druggable targets.

The initial focus of this joint research will be on HER2-low tumors (cancers that express low levels of the growth-promoting protein HER2), a historically challenging clinical area, with the potential to expand into additional tumor types based on data outcomes and joint scientific discussions. By layering spatial context over proteome-level data, the teams aim to pinpoint cell surface proteins that are uniquely positioned for emerging therapeutic modalities, including antibody-drug conjugates and cell-based therapies.
This sophisticated AI-driven spatial multimodal and deep proteomics pipeline is spearheaded by Tae Hyun Hwang, PhD, professor of Surgery, founding director of Molecular AI Initiative and director of AI Research in the Section of Surgical Sciences at Vanderbilt Health. Hwang also co-leads gastric cancer atlas efforts within the National Cancer Institute-funded Human Tumor Atlas Network (HTAN) and is spearheading international HTAN collaborations with South Korea’s National Cancer Center.
Highlighting the clinical necessity of this integrated approach, Hwang said, “Identifying therapeutic targets and understanding treatment response require a precise view of proteins, spatial context and tumor biology. By combining Vanderbilt Health’s Molecular AI and spatial analysis capabilities with Bertis’ proteomics and AI-enabled target discovery platform, this collaboration is designed to generate high-confidence therapeutic targets and predictive biomarkers that can support future translational research and therapeutic development.”
Bertis is led by co-CEOs Dong-young Noh and Seung-man Han, who emphasized the collaboration accelerates the global reach of their platform.
“Collaborating with Vanderbilt Health, a leading U.S. academic medical center with strong expertise in Molecular AI, spatial biology and cancer research, is highly meaningful and reflects the growing global recognition of Bertis’ technological capabilities,” Han said. “Through this collaboration, we aim to expand the role of AI-driven proteomics in drug discovery and identify therapeutic targets that may open new possibilities in oncology.”
The post Vanderbilt Health and Bertis establish collaboration for cancer drug discovery appeared first on Vanderbilt Health News.




